
Water holding capacity - how cellulose fibrils does it
Anni Karppinen | December 12, 2017
Water holding capacity, or high water retention value, is often mentioned as a key property of cellulose fibrils. When it is dispersed into water, the fibrils trap water between them and do not release it easily. As a consequence, even rather low concentration of MFC in water has gel-like appearance since the water is not able to flow freely. What is behind this? Let’s try to find out.
Large surface area and high amount of hydroxyl groups
Cellulose fibrils has a huge surface area which makes it possible to bind a lot of water to the fibrils. This large surface area comes from the fibrillation process where the raw material, some kind of cellulose fibers, is torn down to thinner and thinner fibrils. At the same time, the surface area grows.
It is challenging to measure the surface area precisely, but it has been estimated to be 100-200 m2/g (Lavoine et al. 2012) which means that 1 g of cellulose fibrils has the same surface area as a three-bedroom house – all of it covered with water molecules.
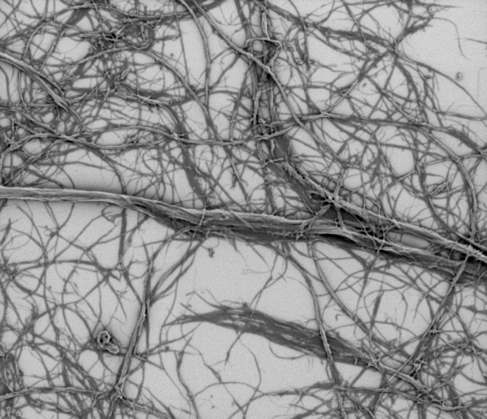
Cellulose fibrils consists of thin and long fibrils with high surface area
Another important reason for the high water holding capacity is the special interactions which the cellulose fibril surface has with water. The fibers mainly consists of cellulose and the cellulose chain has a lot of hydroxyl (OH) groups. These OH groups form strong hydrogen bonds with water molecules and keeps them bound on the fibril surface. For those who are interested in details of the cellulosic surface, I recommend to read an article by Hubbe and Rojas (2008).
Not an absorbent
Although cellulose fibrils is excellent at holding water, it is not an absorbent. In other words, after drying, MFC does not absorb the same amount of water. Drying reduces the surface area of the fibril and changes its surface character permanently. This change is called hornification and occurs on all cellulosic surfaces. Hornification of pulp fibers was described for the first time in the 1940’s by G. Jayme and has been studied quite extensively. For instance, it has been shown that certain polymers can improve the redispersing of dried cellulose fibers. If you want to know more about the scientific background, a summary by Fernandes Diniz and colleagues is a good starting point.
By the way, hornification is an important process, since it facilitates another key character of cellulose fibrils; the excellent film forming ability.
→ Read also: What is MicroFibrillated Cellulose (MFC)?
How to benefit from it?
We've learned that the huge size and, even more, the special nature of the surface area, makes the cellulose fibrils so good at holding water. Water holding ability is closely related to many applications of cellulose fibrils. For example, stabilizing water based formulations, keeping a surface longer wetted, moisturizer in cosmetic products and controlling the drying of coatings. In these kinds of applications it can replace synthetic performance additives produced from oil and contribute to greener products.
Editor's note: This post was originally published in April 2016. We've revamped and updated it for accuracy and comprehensiveness.
Written by:
Anni Karppinen
